How are electrons arranged?
- the first energy level (closest to the nucleus) holds a maximum of two electrons
- the second energy level holds a maximum of eight electrons
- electrons are paired in twos, this represents the filling of orbitals
- each energy level has different orbitals, and each orbital can hold two electrons
- any unpaired electron is always in the outermost energy level
- for main group elements, lower energy levels fill completely before electrons are placed into the next level
- main group: elements in the s and p blocks of the periodic table
- for main group elements, lower energy levels fill completely before electrons are placed into the next level
Shells (energy levels)
- quantized energy levels that electrons can occupy
- electrons can not exist at energies between levels
- labeled starting from the nucleus
- the higher the shell number, the greater the energy of electrons in that shell
- the shells represent electrons’ energy levels, NOT their position or path
- electrons do not move in circular paths around the nucleus
- sometimes, the Bohr model may show rings that get closer together as the shell number increases
- represents how the difference between energy levels decreases with greater
- exact spacing of the rings is not important
- maximum number of electrons per shell is determined by the formula:
- electrons in the outermost shell are called valence electrons
| Shell ⇅ | Max electrons ⇅ |
|---|---|
| 2 | |
| 8 | |
| 18 | |
| 32 | |
| … | … |
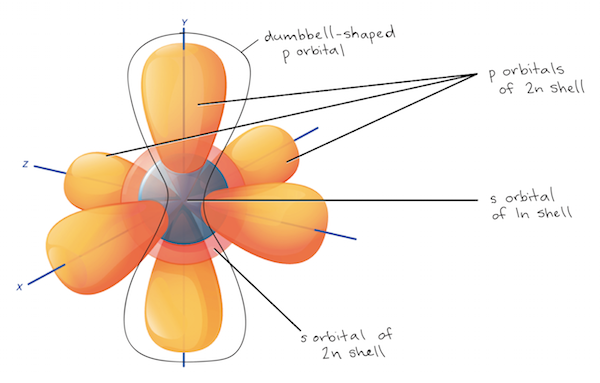
Subshells, and orbitals
- subshells — the different configurations/shapes of a shell
- composed of orbitals
- orbitals — each unique orientation of the subshells
- each orbital can hold up to 2 electrons
- specifies the shape and position of the regions that electrons occupy
| Subshell label ⇅ | Max electrons ⇅ | In shells ⇅ | Shape ⇅ |
|---|---|---|---|
| (s)harp | 2 | every shell | 1 single, spherical orbital |
| (p)rincipal | 6 | 2nd shell+ | 3 dumbbell-shaped orbitals, at right angles |
| (d)iffuse | 10 | 3rd shell+ | 5 orbitals, complex |
| (f)undamental | 14 | 4th shell+ | 7 orbitals, complex |
| (g) | 18 | 5th shell+ | 9 orbitals (theoretically) |
Electron configuration notation
- to explain how an atom’s electrons are arranged in orbitals
- written as the element symbol with subshell labels superscripted with the number of elections in each subshell
- ex.
- can be condensed by building upon the most previous noble gas, instead of listing every shel, subshell, and electron count
- ex. etc.
- for ions, exactly the same but add/remove electrons to/from the highest energy orbital
- atoms/ions that have the same electron configuration are called isoelectronic
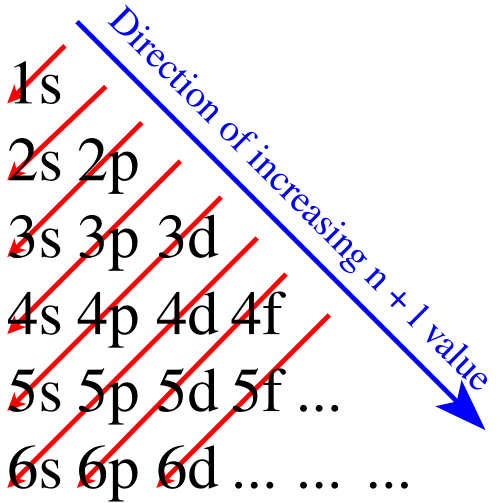
Aufbau principle
- German for “building-up” principle
- electrons first fill up subshells of the lowest available energy, to form the most stable electron configuration possible
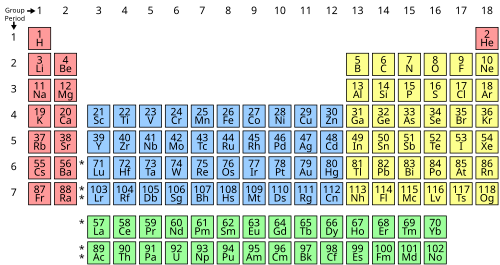
- the elements in the periodic table can be broken up into “blocks” that group them by the orbitals their valence electrons fill
- only accurate for the first 20 elements, up to calcium, then does not accurately predict electron placement