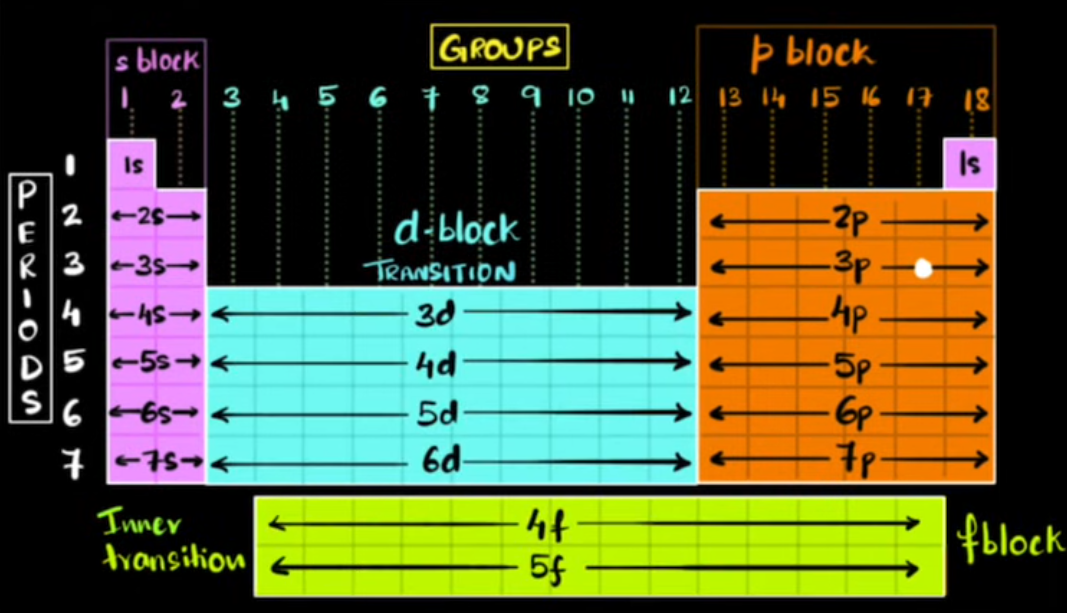
Periods, groups, and blocks
Period
- row in the periodic table
- each element in a period has the same number of shells
Group
- column in the periodic table
Block
- set of related elements
- each element has its valence electrons in the same orbital
d-block
- “transition” elements
- electrons are never added to the valence shell
- period - 1 = subshell that electrons are placed in
f-block
- “inner transition” elements
- similar to d-block, electrons are not added to the valence shell
- period - 2 = subshell that electrons are place in
Links