- atoms can receive external energy via heat, electricity, and/or EM radiation (light/photons)
- when an electron absorbs a photon with energy equal to the difference between its current energy level and another, it enters an excited state and transitions to the higher energy level 1
- if the energy is too high, the electron can escape the atom and the atom becomes ionized
- excited electrons are unstable want to return to the most stable, e.g. the lowest possible, energy level, called the ground state
- ground state — lower energy, stable
- excited state — higher energy, unstable
- when an electron transitions from a higher energy level to a lower one, it emits a photon
- the energy/frequency of the emitted photon is determined by the difference in energy between the two levels the electron transitioned between
- each element has unique discrete energy levels where its electrons can exist
- due to having different numbers of protons and electrons
- because of each elements’ unique energy levels, the light emitted by atoms of each element will have a distinct combination of frequencies
- this unique signature of frequencies is the atomic spectra of an element
- by analyzing the atomic spectra of some body [%, like a planet or star, we can determine its elemental composition
- this is called spectral analysis
- molecules also have unique sets of energy levels, and therefor unique spectra
Energy levels

- atoms have an infinite number of energy levels, but a finite amount of energy is required for an electron to escape its atom
- an energy level diagram helps predict the photon energies by an atom
Two types of spectra
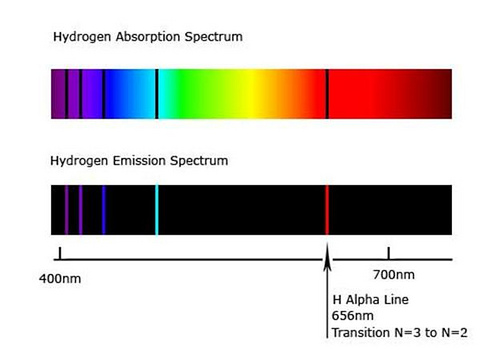
Absorption spectra
- light from another source (a star) passing through cooler gas (like a planet’s atmosphere) 2
- the cooler atoms absorb the light, but when the electrons transition down and emit their photons, the emitted photons are scattered in random directions
- the light that was travelling from the source to us/Earth, will have fewer of the specific frequencies absorbed/emitted
Emission spectra
- light directly emitted from excited atoms
- only the specific photon frequencies from electrons transitioning from higher to lower energy levels
Tools
- spectroscope — uses a diffraction grating to analyze the spectrum of a light source
Footnotes
-
TODO — what happens if an atom receives multiple types of energy at the same time? eg. if an electron normally requires energy from an 100nm frequency photon to move from to , if the atom is heated up and receives some smaller frequency photon (<100nm), can the heat energy combine with the photon energy and transition the photon from to as well, or does the energy have to the same type? ↩
-
TODO — can liquids/solids produce atomic spectra? ↩